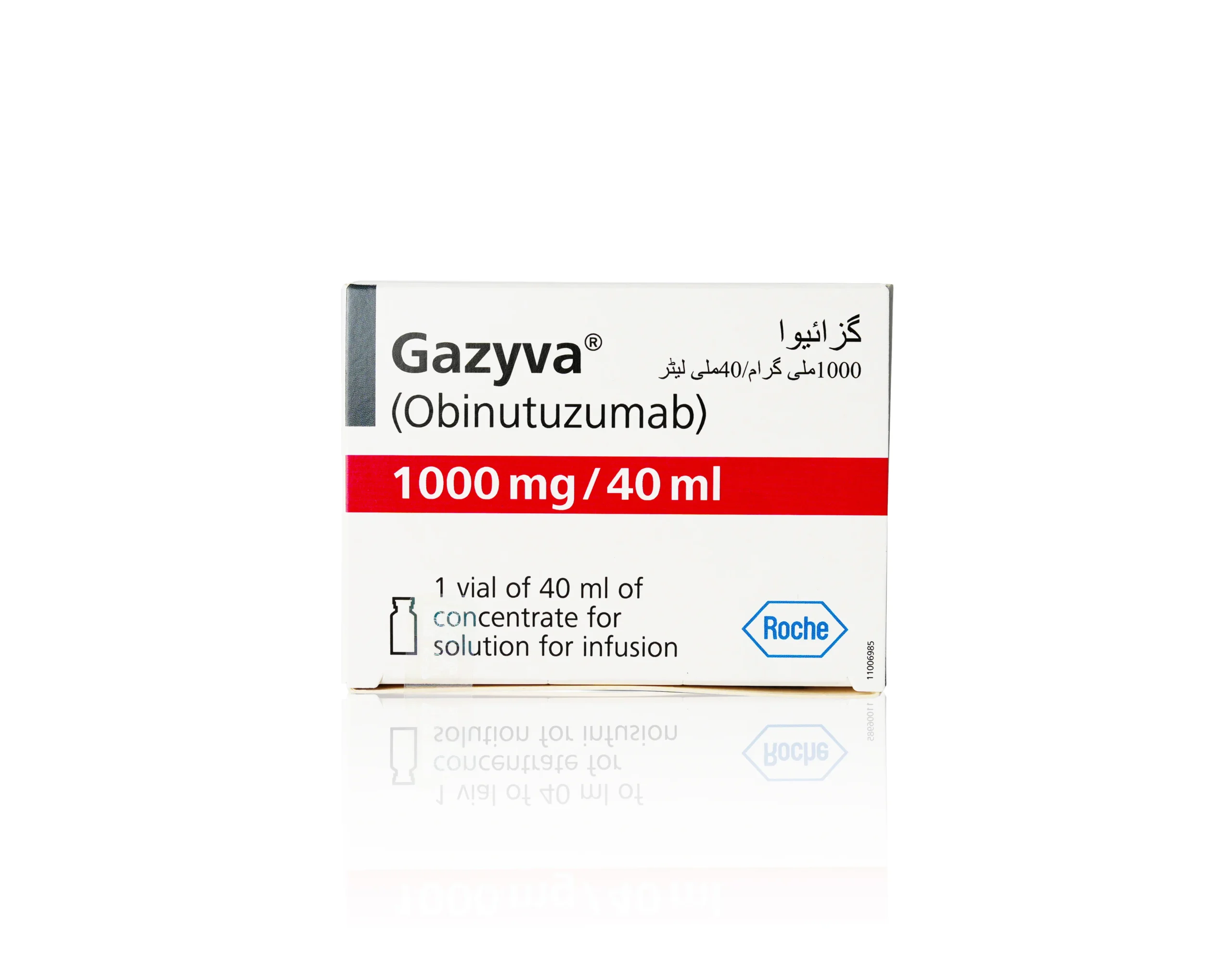
Gazyva (Obinutuzumab) 1000mg
Grazyva is a crucial medicine for Chronic Lymphocytic Leukemia and Follicular Lymphoma treatment. Gazyva is a Order Gazyva at PakMeds with a valid haematologist or oncologist prescription.
Gazyva is a prescription CD20-directed cytolytic antibody containing Obinutuzumab, used to treat previously untreated chronic lymphocytic leukemia and follicular lymphoma in adults. It was the first medicine approved under the FDA’s Breakthrough Therapy Designation program, receiving initial FDA approval in November 2013. Each single-dose vial delivers 1,000 mg of Obinutuzumab in 40 mL of preservative-free concentrate for intravenous infusion.
| Product Name | Gazyva |
|---|---|
| Manufacturer | Genentech, Inc. (a member of the Roche Group), South San Francisco, CA, USA |
| Active Ingredient | Obinutuzumab (humanized type II anti-CD20 IgG1 monoclonal antibody) |
| Strength | 1,000 mg / 40 mL (25 mg/mL) |
| Formulation | Concentrate for Solution for Intravenous Infusion |
| Pack Size | 1 Single-Dose Vial |
| FDA Approval | November 2013 |
| Prescription Required | Yes |
| Generic Name | Obinutuzumab |
Gazyva Ingredients and Usage
Gazyva contains Obinutuzumab, a humanized type II anti-CD20 monoclonal antibody of the IgG1 subclass. It is produced by mammalian cell (CHO) suspension culture and glycoengineered with reduced fucose content in its Fc region — a structural modification that significantly enhances immune effector cell activation compared to first-generation anti-CD20 antibodies such as rituximab. Each 40 mL vial is formulated in L-histidine, L-histidine hydrochloride monohydrate, trehalose dihydrate, and poloxamer 188 at pH 6.0.
Gazyva is indicated for three cancer types. In combination with chlorambucil, it treats previously untreated chronic lymphocytic leukemia (CLL) in adults. In combination with bendamustine followed by Gazyva monotherapy, it treats follicular lymphoma (FL) that relapsed after or is refractory to a rituximab-containing regimen. In combination with chemotherapy followed by Gazyva monotherapy, it treats previously untreated stage II bulky, III, or IV follicular lymphoma in adults who achieve at least a partial remission to induction.
Each dose is 1,000 mg administered by slow intravenous infusion through a dedicated line. For CLL, the first 1,000 mg is split across Day 1 (100 mg) and Day 2 (900 mg) to reduce infusion-related reaction risk. Patients with FL who respond to induction continue Gazyva 1,000 mg monotherapy for up to 2 years. Gazyva must never be given as an IV push or bolus. Premedication with a corticosteroid, antihistamine, and acetaminophen is mandatory before every infusion.
How Does Gazyva Work?
CD20 is a protein antigen expressed on pre-B and mature B lymphocytes, including the malignant B cells responsible for CLL and follicular lymphoma. Gazyva’s Obinutuzumab antibody binds directly to the CD20 antigen and destroys these B cells through three simultaneous mechanisms. It engages natural killer cells and macrophages via its FcgRIII receptor — producing stronger antibody-dependent cell-mediated cytotoxicity (ADCC) than rituximab. It directly activates intracellular death signalling pathways via a reactive oxygen species-dependent mechanism. It also triggers activation of the complement cascade, adding a further layer of tumour cell destruction.
The reduced fucose content of Obinutuzumab’s Fc region is the structural feature that sets Gazyva apart from rituximab. This glycoengineering increases FcgRIII binding affinity and produces superior direct cell death induction in preclinical studies. In the pivotal Phase 3 CLL11 trial, Gazyva plus chlorambucil achieved an overall response rate of 78% versus 65% for rituximab plus chlorambucil. Complete response rate was 21% versus 7%, and progression-free survival was 27 months versus 15 months. In the GADOLIN trial for relapsed or refractory FL, Gazyva plus bendamustine followed by Gazyva monotherapy significantly extended progression-free survival versus bendamustine alone.
B-cell depletion begins rapidly after the first Gazyva infusion. Initial CD19 B-cell recovery is observed in some patients approximately 9 months after the last dose, with B-cell counts returning toward normal over 18 months of follow-up.
Gazyva Side Effects
Gazyva causes significant infusion-related reactions, particularly with the first dose, and produces myelosuppression across all indications. Premedication is mandatory before every infusion. The most commonly reported adverse reactions from the CLL11, GADOLIN, and GALLIUM clinical trials are listed below.
- Infusion-related reactions — occurred in 65% of CLL patients and 60% of previously untreated FL patients during the first infusion; symptoms include nausea, fatigue, chest discomfort, dyspnoea, dizziness, hypotension, flushing, headache, fever, and tachycardia
- Neutropenia — Grade 3 to 4 low white blood cell count, reported in more than 10% of patients across indications
- Thrombocytopenia — low platelet count causing bruising and prolonged bleeding risk
- Anaemia — low red blood cell count causing fatigue, pallor, and shortness of breath
- Fever (pyrexia)
- Cough and upper respiratory tract infections
- Fatigue and generalised weakness (asthenia)
- Nausea, diarrhoea, constipation, and vomiting
- Decreased appetite
- Joint and muscle pain (arthralgia and myalgia)
- Sinusitis and urinary tract infections
- Elevated liver enzymes (ALT and AST), particularly concurrent with infusion reactions or tumour lysis syndrome
- Disseminated intravascular coagulation (DIC) — rare but fatal cases reported, predominantly within 1 to 2 days of the first infusion
Warnings
- BLACK BOX WARNING — Hepatitis B Virus (HBV) reactivation: Gazyva carries an FDA Boxed Warning for HBV reactivation. Reactivation can result in fulminant hepatitis, hepatic failure, and death. It has occurred in patients who are HBsAg positive, in patients who are HBsAg negative but anti-HBc positive, and in patients with apparently resolved hepatitis B infection. All patients must be screened for HBV infection before starting Gazyva. HBV-positive patients require antiviral prophylaxis and close monitoring during and after treatment. Gazyva and all concomitant medications must be discontinued immediately if HBV reactivation occurs.
- BLACK BOX WARNING — Progressive multifocal leukoencephalopathy (PML): Fatal PML caused by reactivation of the JC virus has been reported in patients receiving Gazyva. PML is a serious brain infection that can cause death or severe permanent disability. Patients must be monitored for new or worsening neurological symptoms throughout treatment. Gazyva must be permanently discontinued and expert neurological evaluation initiated if PML is suspected.
- Contraindication — hypersensitivity: Gazyva is contraindicated in patients with a known hypersensitivity reaction — including anaphylaxis or serum sickness — to Obinutuzumab or to any excipient in the formulation.
- Severe infusion-related reactions: Life-threatening and fatal infusion-related reactions can occur during or within 24 hours of Gazyva infusion. Grade 3 to 4 infusion reactions were reported in 20% of CLL patients receiving the first 1,000 mg. Vital signs must be monitored throughout every infusion. Grade 4 reactions require immediate permanent discontinuation. Grade 3 reactions require infusion interruption and restart at no more than half the previous rate after symptom resolution.
- Tumour lysis syndrome (TLS): Fatal TLS has been reported in CLL and NHL patients receiving Gazyva. As Gazyva rapidly destroys cancer cells, their intracellular contents are released into the blood, causing dangerous electrolyte imbalances and potential renal failure. Prophylactic hydration and anti-hyperuricemic agents must be given to patients at high risk of TLS before the first dose. Blood counts and chemistries must be monitored closely.
- Severe myelosuppression: Gazyva causes clinically significant neutropenia, thrombocytopenia, and anaemia. Blood counts must be monitored regularly throughout treatment. Dose interruption or modification is required if severe cytopenias develop. Patients must contact their doctor immediately if they develop fever, unusual bleeding, or signs of infection.
- Disseminated intravascular coagulation (DIC): Fatal and severe DIC has been reported in CLL and FL patients receiving Gazyva, typically within 1 to 2 days of the first infusion and often in association with infusion reactions or TLS. Coagulation parameters, platelet counts, and signs of bleeding or thrombosis must be monitored in any patient with suspected DIC.
- Serious and fatal infections: Gazyva substantially suppresses immune function and increases the risk of serious bacterial, fungal, and viral infections, including opportunistic infections. Fatal infections have been reported in patients on Gazyva in combination with chemotherapy. Active serious infections must be resolved before initiating treatment.
- Antihypertensive medicines: Hypotension can occur during Gazyva infusions. Antihypertensive medicines should be withheld for 12 hours before and throughout each Gazyva infusion and for 1 hour after administration.
- Live virus vaccines: Live or attenuated viral vaccines must not be administered during Gazyva treatment or until B-cell recovery is confirmed following the final dose. Infants born to mothers who received Gazyva during pregnancy may have depleted B-cell counts at birth and must not receive live virus vaccines until B-cell recovery is established.
- Pregnancy: Monoclonal antibodies cross the placenta. Based on its mechanism of action, Gazyva can cause B-cell depletion in infants exposed in utero. Animal studies at 2.4 times the clinical dose showed opportunistic infections and immune complex mediated hypersensitivity reactions in offspring, though no direct embryo-toxic or teratogenic effects were observed. Women of reproductive potential must use effective contraception during Gazyva treatment and for 6 months after the final dose.
- Breastfeeding: Human IgG is present in human milk. Because of the potential for serious adverse reactions in a breastfed infant, women must not breastfeed during Gazyva treatment or for 6 months after the final dose.
- Palifermin interaction: Palifermin must not be administered within 24 hours before, during, or within 24 hours after Gazyva infusion, as coadministration increases the severity and duration of mucositis.
Gazyva Storage Conditions
Store unreconstituted Gazyva single-dose vials under refrigeration at 2°C to 8°C (36°F to 46°F). Keep vials in the original carton to protect from light. Do not freeze and do not shake the vials. Do not use beyond the expiry date stamped on the carton.
Once diluted for intravenous infusion into a 250 mL 0.9% sodium chloride infusion bag, the prepared solution must be used immediately for microbiological safety. If immediate use is not possible, the diluted solution may be stored under refrigeration at 2°C to 8°C for up to 24 hours, followed by use at room temperature for up to an additional 24 hours. Discard any unused portion after 24 hours. Mix the diluted infusion bag by gentle inversion only — never shake or freeze the prepared solution. Do not mix Gazyva with any other medicinal product in the same infusion bag or infusion line. Keep all vials out of reach of children at all times.
Reviews
There are no reviews yet.


(0 customer reviews)