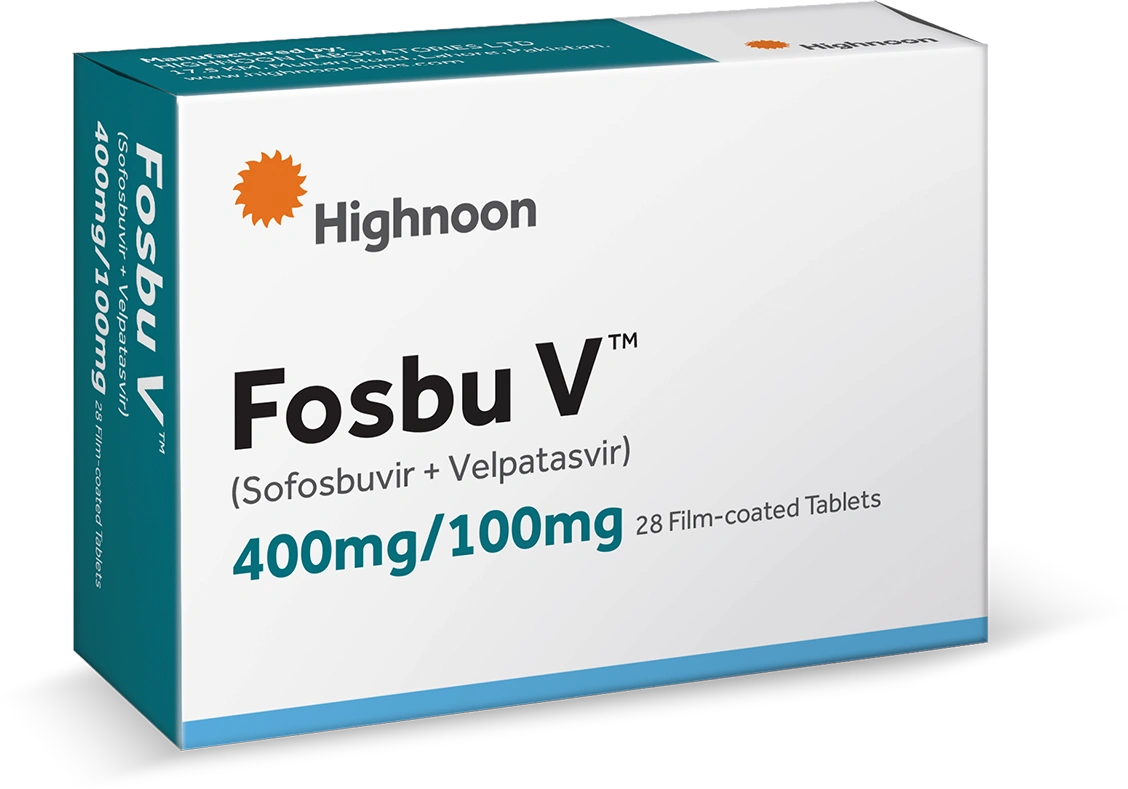
Fosbu V 400/100Mg (28 Tablets)
Buy Fosbu V online at PakMeds and save on genuine, prescription verified medicine. We deliver the medicine to your door across Pakistan.
Fosbu V treats chronic Hepatitis C virus (HCV) infection across all six genotypes with a 95 to 99 percent cure rate. It is a once daily oral tablet taken for just 12 weeks.
| Manufacturer | Highnoon Laboratories Limited |
|---|---|
| Active Ingredient(s) | Sofosbuvir 400mg, Velpatasvir 100mg |
| Strength | 400mg / 100mg |
| Pack Size | 28 Tablets |
| Prescription Required | Yes |
| Generic Name(s) | Sofosbuvir, Velpatasvir |
Fosbu V Ingredients and Usage
Fosbu V contains two direct acting antivirals: sofosbuvir (400mg) and velpatasvir (100mg). Sofosbuvir is a nucleotide analog NS5B polymerase inhibitor. Velpatasvir is an NS5A replication complex inhibitor.
Together, these two ingredients form a pan genotypic regimen for HCV genotypes 1, 2, 3, 4, 5, and 6. The WHO and FDA both approve this combination for adults and children aged 6 years and older. Fosbu V is indicated for patients with or without liver cirrhosis.
For patients with decompensated cirrhosis (Child-Pugh B or C), Fosbu V is prescribed alongside ribavirin. This combination regimen runs for 12 weeks under specialist supervision.
How Does Fosbu V Work?
Fosbu V blocks the Hepatitis C virus at two separate stages of its replication cycle. Sofosbuvir inhibits the NS5B RNA dependent RNA polymerase — the enzyme HCV uses to copy its genetic material. This halts new viral RNA synthesis directly at the source.
Velpatasvir blocks the NS5A protein, which HCV needs for both replication and assembly of new virus particles. Disabling NS5A prevents the virus from spreading further within liver cells. This dual action reduces viral load rapidly and drives the high cure rates seen in clinical trials.
Fosbu V Side Effects
Fosbu V is generally well tolerated. In Phase III trials across 1,558 subjects, severe side effects occurred in only 3 percent of patients. The most commonly reported adverse effects include:
- Headache — reported in up to 22% of patients in ASTRAL-1 trials
- Fatigue or tiredness — reported in approximately 15% of patients
- Nausea — affects around 9% of patients, typically mild and transient
- Asthenia (muscle weakness) — reported in about 5% of patients
- Insomnia — reported in 5% of patients during the treatment period
- Nasopharyngitis (common cold symptoms) — mild upper respiratory irritation
- Diarrhea — more common when co administered with ribavirin
Warnings
- Hepatitis B reactivation: Patients co infected with HBV risk serious HBV flare, liver failure, or death. Screen all patients for HBV infection before starting Fosbu V.
- Amiodarone interaction: Combining sofosbuvir with amiodarone can cause life threatening bradycardia or cardiac arrest. Avoid this combination; if unavoidable, monitor in an inpatient setting for the first 48 hours.
- CYP enzyme inducers: Rifampin, carbamazepine, phenytoin, phenobarbital, and St. John’s Wort reduce velpatasvir plasma levels significantly. Avoid concurrent use to prevent treatment failure.
- Antacids and proton pump inhibitors: Take antacids at least 4 hours before or after Fosbu V. Proton pump inhibitors such as omeprazole reduce velpatasvir absorption and must be timed carefully with food.
- Pregnancy and breastfeeding: Safety in pregnant women has not been adequately established. Breastfeeding while using Fosbu V is not recommended without physician guidance.
- Avoid duplicate sofosbuvir products: Do not use Fosbu V with Sovaldi or any other sofosbuvir containing medicine — this carries a serious overdose risk.
Fosbu V Storage Conditions
- Store below 30°C (86°F) at room temperature — do not refrigerate
- Keep in the original packaging to protect from humidity and light
- Store in a cool, dry place away from direct sunlight and heat sources
- Keep out of reach of children at all times
- Dispose of unused or expired tablets as directed by your pharmacist — do not flush or discard in household waste
Fosbu V — Frequently Asked Questions
How effective is Fosbu V at curing Hepatitis C?
Fosbu V achieves a sustained virologic response (SVR12) in 95 to 99 percent of patients. SVR12 means no detectable HCV in the blood 12 weeks after finishing treatment. This is considered a functional cure by both the WHO and FDA.
Phase III ASTRAL trial data confirmed these results across all six HCV genotypes. Efficacy remains high in patients with compensated cirrhosis. Those with decompensated cirrhosis require a ribavirin combination to achieve comparable outcomes.
How long does a Fosbu V treatment course last?
Most patients take one Fosbu V tablet once daily for 12 weeks. The tablet can be taken with or without food at the same time each day. Do not skip doses — missing doses lowers drug levels in the blood and reduces cure rates.
Patients with decompensated cirrhosis may need 12 weeks of Fosbu V plus ribavirin. Your hepatologist will confirm the duration based on genotype, viral load, and liver condition. Do not stop treatment early without medical advice.
Which Hepatitis C genotypes does Fosbu V treat?
Fosbu V is a pan genotypic regimen — it treats HCV genotypes 1, 2, 3, 4, 5, and 6 with a single tablet. This removes the need for genotype specific treatment planning in most patients. It is one of the few regimens approved by the WHO and FDA for all six genotypes.
Genotype 3 patients, historically the hardest to treat, still achieve cure rates above 95 percent with Fosbu V. Patients co infected with HIV can also use Fosbu V safely. Discuss all co infections and current medications with your doctor before starting treatment.
Reviews
There are no reviews yet.



(0 customer reviews)